Sequence alignment
Pairwise alignment
A) DOT PLOT
The easiest way to align two sequences is to use a dotplot. In its most straight forward implementation the two sequences to be aligned are written along the coordinate axis.
In more realistic implementations a window of 5 to 20 nucleotides or amino acids is slid along one of the axes (i.e., sequences) and compared to every possible window on the other axis (sequence). The dot intensity is adjusted to reflect the percent identity (or similarity) in the two windows. See the dotlet help pages.
![]()
Optimal global and local alignments.
There are many different algorithms to calculate pairwise sequence alignments. For two sequences it is "easy" to calculate an optimal global alignment. (According to the motto: "It can be easily shown" -- see here). The so called Needleman-Wunsch algorithm is widely used, it optimizes a positive alignment score, a related (and under some conditions equivalent approach) is to minimize the differences between to sequences.
![]()
Multiple Sequence Alignments
![]()
Progressive alignment of multiple sequences
|
CLUSTAL, CLUSTALW and CLUSTALX
Usually global alignments are the easiest to calculate (local see discussion of blast )
One of the easiest to use, most sophisticated, and most versatile alignment programs is clustalw
Clustalw runs on all possible platforms (unix, mac, pc), and it is part of most multiprogram packages, and it is also available via different web interfaces.(Higgins DG, Sharp PM (1988) CLUSTAL: a package for performing multiple sequence alignment on a microcomputer. Gene 73:237-244;
Thompson, J.D., Higgins, D.G. and Gibson, T.J. (1994). CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Research, 22, 4673-4680).
Clustalw uses a very simple menu driven command-line interface, and you also can run it from the command line only (i.e. it is easy to incorporate into scripts.)
Clustalx uses the same algorithms as clustalw. However, it has a much nicer interface, it displays information on the level of similarity, and it uses color in the alignment. Especially for amino acids the use of color greatly enhances the ability to recognize conservative replacements. Clustalx2.1 is available for different platforms at the ebi's ftp site (follow your platform, clustalx is stored in the clustalw folders)
Clustal reads and writes most formats used by different programs. The easiest format is the FASTA format:
>
name of sequence or any other information goes in the first line. This line starts
with ">". The line can be longer than 80 characters. The first line
ends with the first paragraph sign.p
The second line contains the sequence
itself; numbers and other non standard characters
are ignored. Be careful if you download sequences. Often the transfer
programs introduce paragraph signs every 100 characters, and the end of a command
line frequently ends up as the beginning of the sequence.
All sequences to
be read should be in a single file.
(sample clustalw input file)
(sample clustalw output file)
Clustal
also reads aligned sequences. If you input aligned sequences you can go directly
to the tree section.
!! Be careful if you make a mistake, and the sequences are
not aligned, your tree will look strange!!
!!! ALWAYS CHECK YOUR ALIGNMENT!!!
Clustal also is useful to reformat and edit alignments, it is very forgiving in reading formats, e.g., you can open the clustal format (*.aln) in a text editor and delete columns and reload the file into clustalw, and output it in the other formats available.
For calculating an alignment, you can select different substitution matrices, and gap penalties (end-gaps can be considered differently!)
Clustal is better than its reputation. It is doing a great job in handling gaps, especially terminal gaps, and it makes good use of different substitution matrices.
To align sequences clustal performs the following steps (aka as progressive alignment*):
1) Pairwise distance calculation
2) Clustering analysis of the sequences
3) Iterated alignment of two most similar sequences or groups of sequences.
It is important to realize that the second step is the most important. The relationships found here will create a serious bias in the final alignment. The better your guide tree, the better your final alignment. You can load a guide tree into clustal. This tree will then be used instead of the neighbor joining tree calculated by clustalw as a default. (The guide tree needs to be in normal parenthesis notation WITH branch lengths).
* the first effective multiple sequence alignment program was developed by Feng and Doolittle
![]()
Other
programs often used for multiple sequence alignment
(We
will not use
these program in this course; if you are already confused by the information provided,
skip to the assignments):
A program available via the www is SAM (sequence alignment and modeling system) by Richard Hughey, Anders Krogh, Christian Barrett, & Leslie Grate at UCSC. The input consists of a multiple sequence file (aligned or not aligned) in FASTA format. The program uses secondary structure predictions, neighboring sites, etc. to place gaps. The program can be accessed at https://compbio.soe.ucsc.edu/sam.html
If your sequences are not very similar, and if you are not able to generate a trustworthy multiple sequence alignment, you can calculate distance trees based on pairwise alignments only. The best program for this purpose is statalign from Jeff Thorne (Thorne JL, Kishino H (1992) Freeing phylogenies from artifacts of alignment. Mol Bio Evol 9:1148-1162). It runs under standard UNIX. It's only worth your effort if you are getting gray hairs because of a data set you cannot reliably align. Very out of fashion these days.
MUSCLE is the current alignment program of choice. It is thought to give better alignments compared to clustal, it is faster and works with larger datasets. The program is available through a webserver at the ebi, and as a commandline program to download here.
Most multiple sequence alignment programs produce alignments that are pleasing to the human eye by placing only a few large gaps into the sequences. However, for many applications it is better to align a particular amino acid to gaps in the other sequences, if one is not certain about the homology of the position. These programs that introduce more gaps are at present underutilized. An example is PRANK (a web version of PRANK is part of the guidance 2 server to determine reliably aligned sites).
In case of divergent sequences, a popular program that combines phylogenetic reconstruction and multiple sequence alignment is SATe.
If you need to use MSA in your work, the current recommendation is to use muscle, and test if an alignment calculated under PKANK gives similar results. Also, if you use less than 100 sequences, try SATe2. (Note the time needed for computation is very different!).
In order to avoid artifacts reflecting the guide tree used for the alignment, many prefer to filter the alignment using only sites that are reliably aligned. One such approach is GBLOCK (implemented in seaview, see below, web server is here), another is guidance from Tal Pupko's lab at TAU (I like this one, because it allows to remove positions from poorly aligned individual sequences, not only complete columns).
Alignments
by Eye:
Jalview (see computer-lab 9) remains a popular option. Advantages are the PCA analysis, and the integration of the different display windows.
Another
useful sequence editor is seaview. It includes more sophisticated programs to align sequences, and to reconstruct molecular phylogenies, and runs on PC and most unix flavors (including Macs). The latest version (4.6) includes phylogenetic reconstruction using phyml and parsimony, multiple sequence alignment using clustalo and muscle, and filtering of poorly aligned regions using GBlocks.
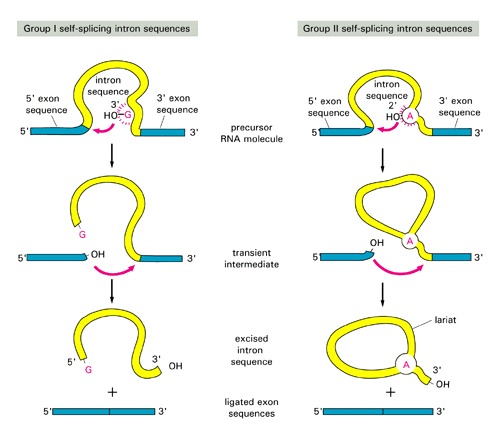
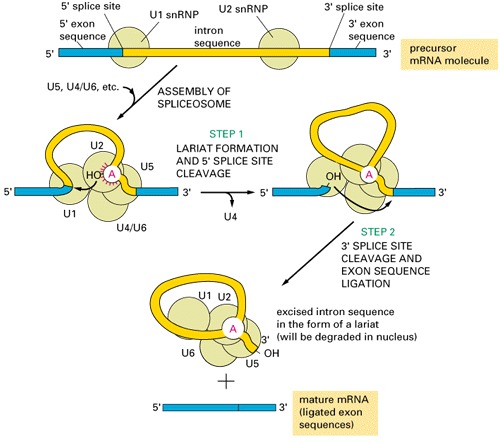
Go over the splice site consensus in Arabidopsis here.